|
12/24/2023 0 Comments Sulfur charge number 
SO3 lewis structure, first, we calculate Q. It acts as a solvent for the manufacture of H 2SO 4. Sulfuric acid is formed when SO3 is combined with water. How to find the formal charge? SO3 exists in liquid form at room temperature which strongly fumes in the air. The O.S of sulfur in sulfuric acid is +6. What is the Oxidation State of S in H 2SO 4?Ģ (Oxdation state of H) + (Oxdation state of S) + 4 (Oxdation state of O) = 0 It needs only two electrons to complete its octet. 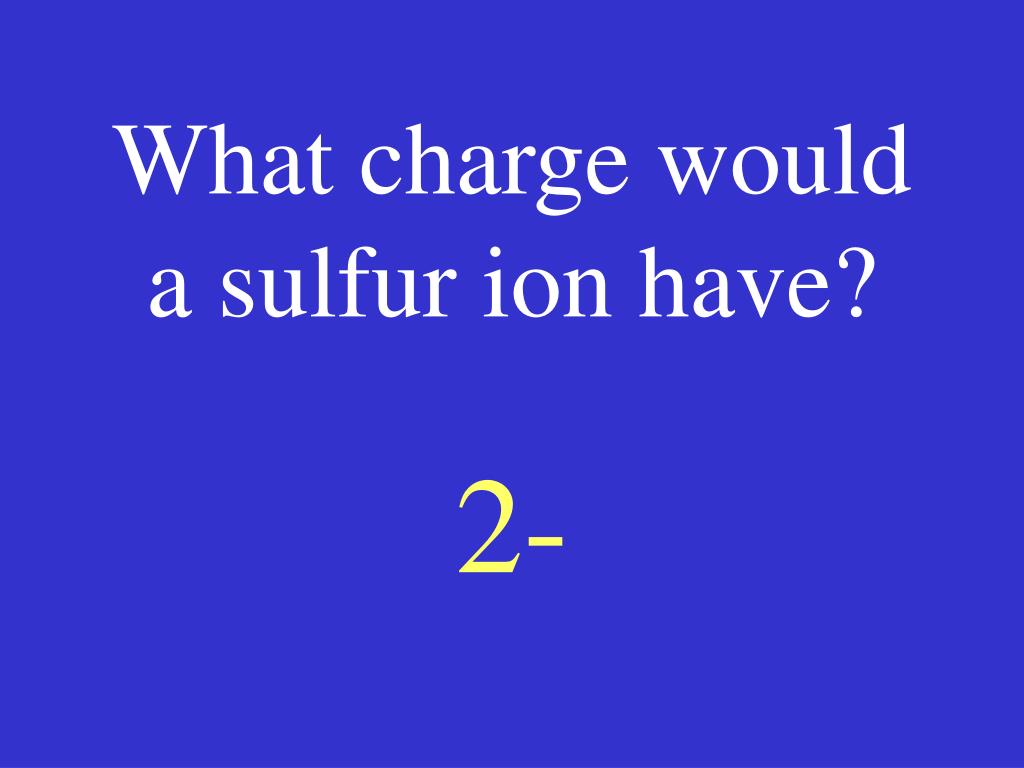
There are 6 electrons present in the outermost shell. The valence shell of the S atom is 3s 2, 3p 4. 
If we look at the electronic configuration of sulfur: Sulfide ( British English also sulphide) 2 is an inorganic anion of sulfur with the chemical formula S 2 or a compound containing one or more S 2 ions. The second method to find the charge is electronic configuration. The charge of O is -2 so all the elements present in the oxygen family have a -2 charge. All the elements present in the oxygen family have the same chemical properties. Period 16 is also called the oxygen family because it is the first element of this period. Sulfur is located in group 3 and in period 16 or 6A just below the oxygen. First of all, you know about the position of sulfur in the modern periodic table. proton number, mass or nucleon number and charge. The first method to find the sulfur charge is by using the periodic table. Sulfur and all compounds of sulfur will be spelled with f, not ph. Only small differences in individual atomic charge of sulfur were observed between lone ion (GP) and ion pair IL(SMD) model systems, indicating that ion–ion interactions do not strongly influence individual atomic charges.Here we discussed the two methods to find out the ionic charge of S. However, the atomic charges of sulfur derived from ChelpG are found to display significant, non-physical conformational dependence. Furthermore, we show that both ChelpG and NBO are reasonable methods for calculating the atomic charge of sulfur in ILs, based on the agreement with both the XPS and NEXAFS spectroscopy results. By comparing the experimental and calculated measures of the atomic charge of sulfur, we provide an order for the sulfur atoms, ranging from the most negative to the most positive atomic charge. Three methods were used to compute the atomic charges: charges from the electrostatic potential using a grid based method (ChelpG), natural bond orbital (NBO) population analysis and Bader’s atoms in molecules (AIM) approach. Atomic charges were computed for lone ions and ion pairs, both in the gas phase (GP) and in a solvation model (SMD), with a wide range of ion pair conformers considered. Our experimental data provides a benchmark for calculations of the atomic charge of sulfur obtained using different methods. In addition, we use Auger electron spectroscopy to show that when XPS binding energies differ by less than 0.5 eV, conclusions on atomic charge should be treated with caution. These values provide experimental measures of the atomic charge in each IL and enable the evaluation of the suitability of NEXAFS spectroscopy and XPS for probing the relative atomic charge of sulfur. The steric number of the sulfur central atom in the SCl4 molecule is 5, thus. Since the overall charge of H2SO3 is 0, and there are two hydrogen atoms, the total oxidation number of hydrogen is +2. charge on nitrogen atom and one double bond between nitrogen and one oxygen atom. Helium, with two protons, will have two electrons. Hydrogen atoms all have one electron occupying the space outside of the nucleus. Since atoms are neutral, the number of electrons in an atom is equal to the number of protons. Solution: Hydrogen typically has an oxidation number of +1. 1: The social security number subatomic-the proton. It is abundant, multivalent and nonmetallic. Experimental near edge X-ray absorption fine structure (NEXAFS) spectra, X-ray photoelectron (XP) spectra and Auger electron spectra are reported for sulfur in ionic liquids (ILs) with a range of chemical structures. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons.The nucleus is composed of protons and neutrons.Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. Determine the oxidation number of sulfur in H2SO3. 32.060.02 (abridged) Sulfur in the periodic table Neon Phosphorus Chromium Arsenic Livermorium abundance Category: Sulfur talk edit references Sulfur (also spelled sulphur in British English) is a chemical element it has symbol S and atomic number 16. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
